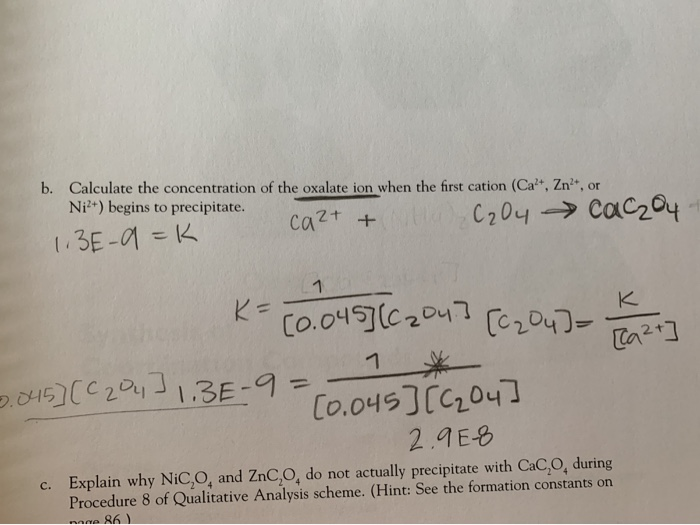Calculate the normality of a solution containing 13.4 g fo sodium oxalate in 100mL solution. - YouTube
Calculation of Lewis basicity (vu) for oxalate anion [(C 2 O 4 ) 2-]... | Download Scientific Diagram

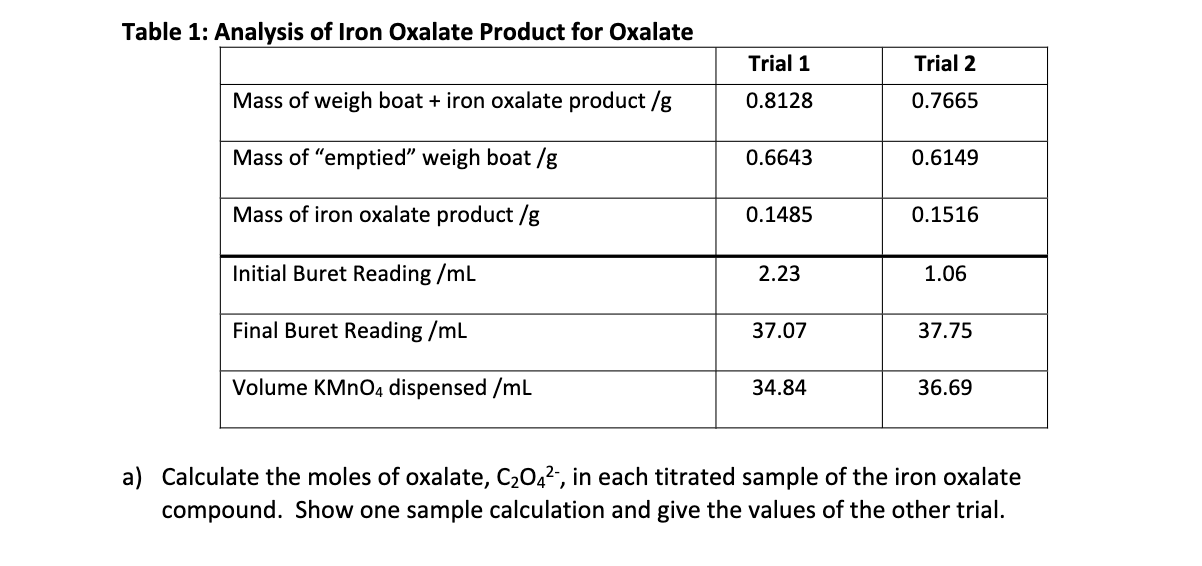
SOLVED: Part B Calculations Determine the % by mass of oxalate (C2042-) in the sample. The reaction between oxalate and permanganate is as follows: 6 Ht + 5 (COOH)z 2 MnO4 =

0.5 g of an oxalate was dissolved in water and the solution made to 100 mL. On titration 10 mL of this solution required 15 mL of (N)/(20)KMnO(4). Calculate the percentage of
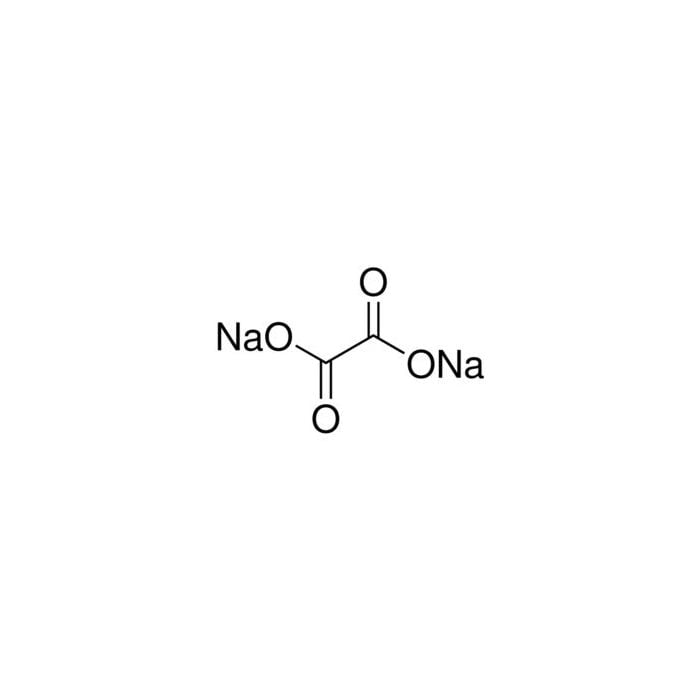
![SOLVED: Calculate the mass percent of oxalate in the following compounds: oxalic acid, sodium oxalate, potassium oxalate, and Na3[Al(C2O4)3] •3H2O. (That dot doesn't mean multiply, it means there are three water molecules SOLVED: Calculate the mass percent of oxalate in the following compounds: oxalic acid, sodium oxalate, potassium oxalate, and Na3[Al(C2O4)3] •3H2O. (That dot doesn't mean multiply, it means there are three water molecules](https://cdn.numerade.com/ask_previews/de5ffcf7-c504-4819-8f25-0d4b402645ee_large.jpg)
SOLVED: Calculate the mass percent of oxalate in the following compounds: oxalic acid, sodium oxalate, potassium oxalate, and Na3[Al(C2O4)3] •3H2O. (That dot doesn't mean multiply, it means there are three water molecules









![Silver Oxalate [Ag2C2O4] Molecular Weight Calculation - Laboratory Notes Silver Oxalate [Ag2C2O4] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2023/03/silver-oxalate-molecular-weight-calculation-300x189.jpg)